|
|
|
Biopolymers
Aquero is interested in biological
polymers, particularly polyamino acids (e.g.,
polyaspartic acid) and the polysaccharides (e.g., starch
and cellulose). These materials, and their building blocks, may be combined to form grafts and blends
with improved properties. We also respect and routinely work with vinyl polymers, which are very capable
molecules, other conventional chemicals, and their interactions with the biological polymers. Background
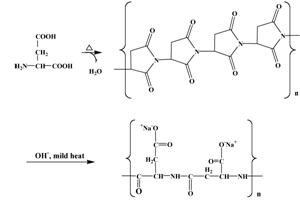
Polyamino acids
The polyamino acids are comprised of amino-acid building blocks, in particular aspartic acid. Also of notable interest to Aquero Company is the amino acid asparagine, a simple variant of aspartic acid. Aspartic acid and its anionic form, aspartate, are very closely analogous to the commodity, vinyl building block, acrylic acid and its anionic form, acrylate. Similarly, asparagine is very closely analogous to the commodity, vinyl building block, acrylamide. Consequently, there arose the aforementioned concept that it might be a good idea to develop replacements for the acrylate/acrylamides, using polyaspartates and copolymers of aspartate and asparagine. Because the markets are so large, the potential reward for developing this new class of biopolymers was also seen as very large and inviting.
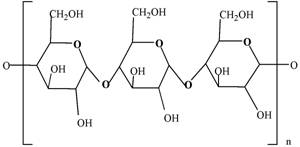
Polysaccharides
Polysaccharides are another major class of biological polymers. The polysaccharides are comprised of simple carbohydrates, or "sugars," or "monosaccharides", as building blocks. Glucose, for example, is the most common of the simple carbohydrates. In fact the polymers of glucose, in particular cellulose, are easily the most abundant molecules in nature. Cellulose makes up cell walls and other structural components of plants and many algae. Starch, the other principal polymer of glucose, is the main storage molecule in all higher plants and again in many algae. Starch and cellulose are relatively cheap and available materials. However, neither are very functional from a strictly chemical point of view. For example, they are not ionic and do not perform like acrylate/acrylamides. Because starch and cellulose are polymeric materials and various reactions could be undertaken to make them ionic, they have been evaluated extensively for markets such as superabsorbents. To date there has been no real success in that effort, due to the low cost and availability of their vinyl counterparts. |
|||||||||||||||||||||||||||||||||